PROMO!
First order? Get 10% OFF with this code: 1storder
Written by
Current research examines whether growth hormone peptides present cancer risks in laboratory studies. Recent studies focus on synthetic substances like CJC-1295 DAC, Ipamorelin, and Sermorelin.
These peptide secretagogues target the pituitary gland to stimulate natural processes, including the secretion of human growth hormone. Scientists investigate potential side effects through controlled clinical trial environments.
Reliable suppliers like Peptide Works provide these compounds exclusively for scientific investigation to researchers worldwide. Understanding these mechanisms helps researchers evaluate long-term safety profiles.
Laboratory analysis reveals how these compounds affect cellular growth patterns. These peptides are for research use only, not for human consumption.
Explore CJC-1295 DAC from Peptide Works, a long-acting growth hormone releasing peptide designed to support cellular regeneration, metabolism, and tissue repair.
Growth hormone peptides influence cellular growth through receptor-mediated activation of the GH/IGF-1 axis. CJC-1295 DAC promotes endogenous growth hormone secretion, and growth hormone activates intracellular signaling pathways (including JAK-STAT) in target tissues, leading to increased IGF-1 production primarily in the liver.
IGF-1 promotes protein synthesis and anabolic responses across multiple cell types. GH/IGF-1 signaling affects chondrocytes involved in cartilage biology and regulates growth plate expansion.
Elevated GH/IGF-1 levels are associated with increased growth responses in laboratory and animal studies. Peptide amino acid sequences determine receptor binding specificity. GH/IGF-1 can interact with immune function, though effects on immune cells vary and remain under investigation. Cellular responses are dose-dependent and exhibit tissue-specific sensitivity to GH/IGF-1 signaling.
Recent studies show elevated IGF-1 levels correlate with certain cancer types. Beneficial effects occur in normal tissue, but concerns exist for malignancies.
Clinical trial data reveals associations with prostate cancer and breast tumors. Research indicates IGF-1, stimulated by Growth Hormone Peptides, promotes cellular division while reducing natural cell death.
Blood tests measuring IGF-1 help assess individual risk factors. Scientists observe this important role in tumor development through multiple pathways.
However, correlation doesn’t prove direct causation in human subjects. Risk of diabetes and other metabolic changes also influence outcomes
Growth hormone peptides influence tumor development primarily through activation of the GH/IGF-1 axis, potentially accelerating existing tumors via JAK-STAT and PI3K pathways in research models. These GH secretagogues indirectly stimulate pathways that promote cellular proliferation.
Studies show CJC-1295 elevates GH/IGF-1 levels without evidence of direct DNA damage through cAMP mechanisms. GH/IGF-1 signaling influences cellular repair and growth processes in laboratory settings.
Sermorelin elevates GH levels by activating the GHRH receptor in research applications. Unlike recombinant human GH, these peptides work through indirect pituitary receptor binding.
Scientists observe enhanced muscle mass alongside monitored changes in cellular proliferation. Research indicates GH/IGF-1 pathways may accelerate existing tumor growth rather than initiating cancer development.
Explore Sermorelin from Peptide Works, a synthetic analog of GHRH that stimulates natural growth hormone production to support vitality, fat metabolism, and sleep quality.
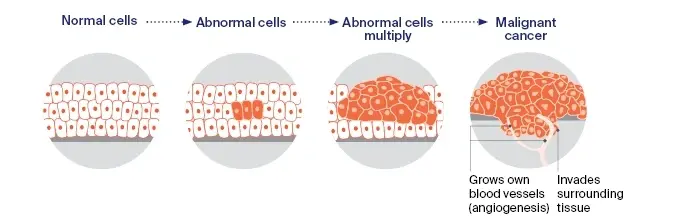
Growth hormone peptides such as CJC-1295 and Ipamorelin do not directly inhibit DNA repair systems. They raise GH levels. Elevated GH can suppress ATM kinase activation in epithelial cell models. GH reduces ATM autophosphorylation, which impairs DNA break signaling.
CJC-1295 raises GH through GHRH receptors. High GH has been shown in epithelial studies to increase TRIM29 and reduce Tip60 activity, which weakens DNA damage response pathways in those models. Ipamorelin elevates GH via ghrelin receptors. Reduced ATM signaling under high-GH conditions is associated with decreased p53 and Chk2 activation, allowing cells to continue proliferating despite damage. GH impairs DNA damage response markers in epithelial studies.
Laboratory tests show GH reduces markers such as phosphorylated H2AX. Evidence for direct Rad50 suppression is limited. Cells retain more unrepaired DNA in GH-treated research models.
Discover Ipamorelin from Peptide Works, a selective growth hormone secretagogue that promotes lean muscle growth, recovery, and anti-aging support with minimal side effects.
P53 failure removes key cell controls in body systems. Growth hormone peptides research shows links between P53 problems and immune system issues.
Cells lose their power to control protein synthesis during repair cycles. This causes wild cell division affecting lean body mass and fat mass balance. CJC-1295 studies show changed amino acid work when P53 paths break down.
Lab tests reveal reduced cognitive function with loss of cell death responses. Blood pressure control gets damaged through these connected failure systems. Research shows P53 loss speeds up cancer growth across tissue types.
Normal cells follow strict death signals when damaged beyond repair capabilities. Cancer cells develop multiple resistance methods that block these natural death commands.
They increase anti-death proteins while reducing molecules that trigger cell suicide. Research shows these cells ignore telomere shortening during the aging process normally.
Studies indicate altered sex hormones and disrupted food intake patterns may influence resistance. Laboratory data reveals modified growth hormone release affects cellular death timing mechanisms.
Cancer cells essentially become immortal by avoiding programmed elimination processes. This resistance allows continued division despite accumulating genetic damage over time
Telomeres release specific biochemical signals when they reach critically short lengths. These molecular timers activate senescence pathways that halt cellular reproduction permanently.
Growth hormone peptides research reveals connections between telomere signaling and beneficial effects on cellular longevity. Short telomeres trigger inflammation cascades that promote tissue breakdown mechanisms.
Studies show these aging signals affect athletic performance through reduced cellular energy production. Research indicates compromised telomeres increase body mass while decreasing synthetic growth hormone effectiveness.
The biochemical clock essentially broadcasts “aging alerts” throughout tissue systems. These molecular warnings coordinate whole-body aging responses across multiple organ systems.
Multiple cellular checkpoints activate permanent division shutdown mechanisms in research models. The Hayflick limit sets maximum division counts through telomere exhaustion pathways.
Growth hormone peptides research shows connections between division limits and weight loss regulation. Laboratory studies reveal DNA damage triggers irreversible senescence through p53 activation.
Cells detect chromosomal instability and activate permanent shutdown responses automatically. Research indicates these triggers affect joint pain development and high blood pressure regulation.
Studies show secretion of growth hormone influences cellular division timing mechanisms significantly. These permanent triggers prevent unlimited cellular reproduction across all tissue types.
The future of growth hormone peptides in cancer research is both promising and complicated. Scientists continue examining how CJC-1295 DAC, Ipamorelin, and Sermorelin interact with growth factor signaling.
Emerging studies aim to balance potential benefits of HGH like body fat mass reduction, muscle mass retention, and enhanced cognitive function, against risks such as prostate cancer and diabetes. This delicate balance is critical for medical doctors, researchers, and ethical supplement development.
Despite public interest in performance enhancement and anti-aging, these compounds remain restricted to research use. Yet their insights continue shaping therapies aimed at better managing gh secretion, body composition, growth hormone secretion, and even cancer itself.
All products discussed are supplied for research purposes only and are not intended for human use.
(1) Jenkins PJ, Mukherjee A, Shalet SM. Does growth hormone cause cancer? Clin Endocrinol (Oxf). 2006 Feb;64(2):115-21.
(2) Cianfarani S. Risk of cancer in patients treated with recombinant human growth hormone in childhood. Ann Pediatr Endocrinol Metab. 2019 Jun;24(2):92-98.
(3) Berlanga-Acosta J, Abreu-Cruz A, Herrera DGB, Mendoza-Marí Y, et al. Synthetic Growth Hormone-Releasing Peptides (GHRPs): A Historical Appraisal of the Evidences Supporting Their Cytoprotective Effects. Clin Med Insights Cardiol. 2017 Mar 2;11:1179546817694558.
(4) Su Y, Dang NM, Depypere H, Santucci-Pereira J, Gutiérrez-Díez PJ, Kanefsky J, Janssens JP, Russo J. Recombinant human chorionic gonadotropin induces signaling pathways towards cancer prevention in the breast of BRCA1/2 mutation carriers. Eur J Cancer Prev. 2023 Mar 1;32(2):126-138.
(5) Boguszewski MCS, Boguszewski CL, Chemaitilly W, Cohen LE, et al. Safety of growth hormone replacement in survivors of cancer and intracranial and pituitary tumours: a consensus statement. Eur J Endocrinol. 2022 Apr 21;186(6):P35-P52.
(6) Muccioli G, Broglio F, Valetto MR, Ghè C, Catapano F, Graziani A, Papotti M, Bisi G, Deghenghi R, Ghigo E. Growth hormone releasing peptides and the cardiovascular system. Ann Endocrinol (Paris). 2000 Feb;61(1):27-31. PMID: 10790589.
ALL CONTENT AND PRODUCT INFORMATION AVAILABLE ON THIS WEBSITE IS FOR EDUCATIONAL PURPOSES ONLY.
DISCLAIMER: These products are intended solely as a research chemical only. This classification allows for their use only for research development and laboratory studies. The information available on our Peptide Works website: https://peptide-works.com/ is provided for educational purposes only. These products are not for human or animal use or consumption in any manner. Handling of these products should be limited to suitably qualified professionals. They are not to be classified as a drug, food, cosmetic, or medicinal product and must not be mislabelled or used as such.
Peptide Works
Related Articles
Vilon Peptide in Organ Research: Cardiac, Renal, and Gastrointestinal Impacts
Vilon peptide demonstrates measurable relevance in organ-focused research models. Researchers investigate this peptide due to its influence on gene activity, immune signaling, and cellular

Copper peptides are small amino acid fragments that bind with copper ions to form biologically active compounds. These molecules have
What are the Best Cognitive Peptides?
Cognitive peptides are short chains of amino acids studied for their potential influence on brain function. Researchers examine how these